Nous utilisons des cookies pour vous offrir la meilleure expérience sur notre site. Vous pouvez en savoir plus sur les cookies que nous utilisons ou les désactiver dans les paramètres de cookies
GINA X CDS - Centralized Data Solution
GINA X is a data acquisition and evaluation software for different analytical instruments used during quality control of radiopharmaceuticals. Simply collect all data from typical Quality Control steps like radiochemical purity, residuals measurement, pH, endotoxin or radio-isotopic purity. Data from instruments such as Radio chromatographs (HPLC & TLC), GC, MCA, LAL and dose calibrator are typical needed to confirm that the products are fully compliant to pharmacopeia or FDA regulations and standards. GINA X will lead you through the entire workflow from the first measurement until signing of the QC analysis certificate. Integrated Methods and Automatic Data Transfer are key features to document the whole process complying with the SOP in a GMP environment.
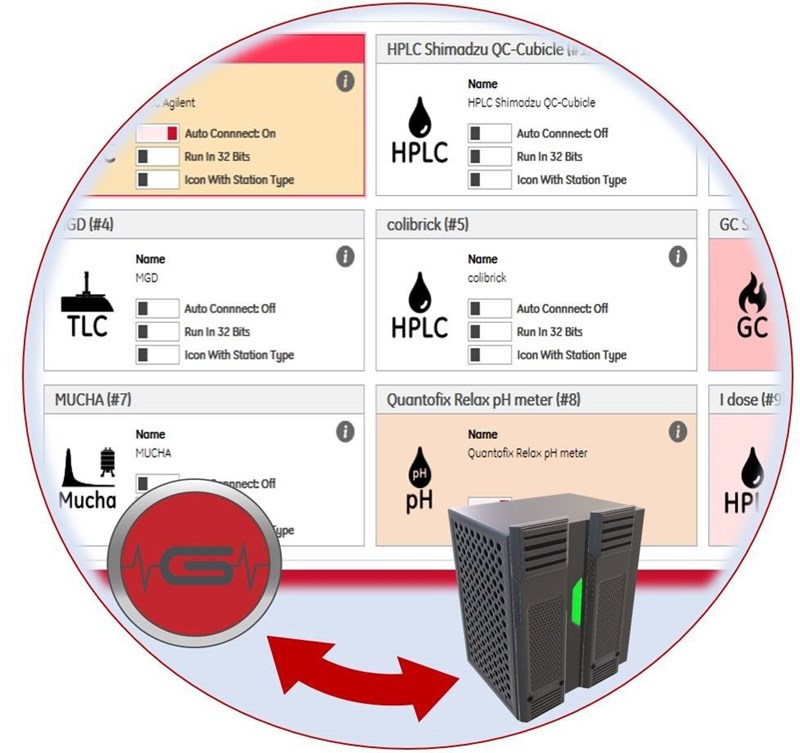
GINA X is the heart of your QC laboratory, offering a single software platform to control all your QC instruments with a unique interface. With the GINA X CDS platform, you are able to control and analyse the data from radio-HPLC’s, all Elysia radio detectors, TLC systems, GC’s, our multichannel analyser and our dose calibrator with a single User Interface. This intuitive User Interface renders additional interfaces unnecessary. Automatic Data Transfer and verification of other QC instruments like endotoxin, pH, osmometry or others is also possible.
The centralized solution is based on a SQL database. The client/server architecture allows an easy centralization and access to the instruments and data from different PC’s. With all data stored in the central SQL database, it is easy to secure the data integrity and be fully GMP and 21CFR part11 compliant.